
Abstract
Glutamine release to the circulation and availability is mainly controlled by key metabolic organs, such as the gut, liver, and skeletal muscles. During catabolic/hypercatabolic situations glutamine can become essential for metabolic function, but its availability may be compromised due to the impairment of homeostasis in the inter-tissue metabolism of amino acids. For this reason, glutamine is currently part of clinical nutrition supplementation protocols and/or recommended for immune suppressed individuals. However, in a wide range of catabolic/hypercatabolic situations (e.g., ill/critically ill, post-trauma, sepsis, exhausted athletes), it is currently difficult to determine whether glutamine supplementation (oral/enteral or parenteral) should be recommended based on the amino acid plasma/bloodstream concentration (also known as glutaminemia). Although the beneficial immune-based effects of glutamine supplementation are already established, many questions and evidence for positive in vivo outcomes still remain to be presented. Therefore, this paper provides an integrated review of how glutamine metabolism in key organs is important to cells of the immune system. We also discuss glutamine metabolism and action, and important issues related to the effects of glutamine supplementation in catabolic situations.
Keywords: amino acids; gut; leukocytes; liver; nutrition; skeletal muscle.
Conflict of interest statement
Figures
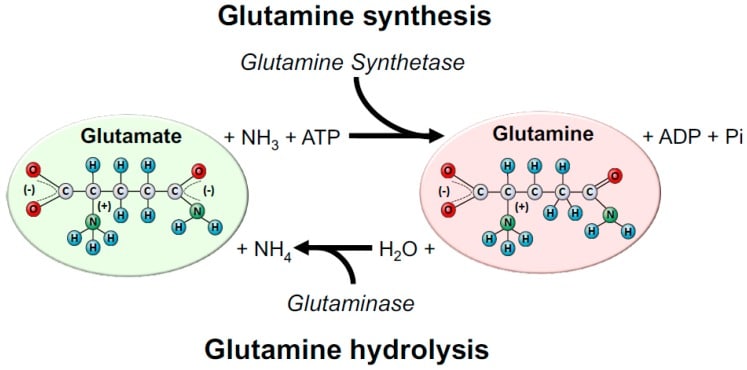
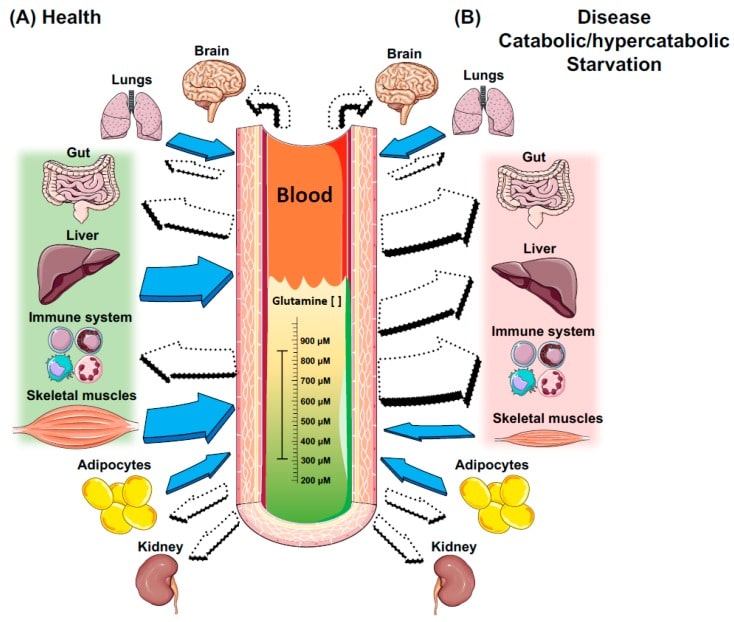
Figure 2: Intertissue glutamine production and utilisation in health and catabolic/hypercatabolic situations. Filled arrows indicate tissues that exhibit GS activity and thus produce glutamine; white arrows indicate tissues that exhibit GLS activity, and thus consume glutamine. In health and/or fed states, glutamine stores are in equilibrium in plasma/bloodstream and tissues, and are maintained constantly mainly by the liver and skeletal muscles, two major stores of glutamine in the body. On the other hand, cells of the immune system are extremely dependent on glucose and glutamine in situation (A), and even more in situation (B). Although the gut is a major site of glutamine consumption, in situation (B), there is a dramatic increase in glutamine consumption from both the luminal and basolateral membrane, when compared to situation (A). In addition, the liver switches the role of a major producer to a major glutamine consumer to maintain gluconeogenesis, and the whole body relies on the skeletal muscle’s ability/stores to maintain glutamine levels. However, this process is usually accompanied by a dramatic increase in muscle proteolysis, atrophy, and cachexia. The lungs and adipose tissue exhibit both GS and GLS enzymes, and hence can produce and consume glutamine in situations (A) and (B). The brain and the kidneys do not exhibit GS, only GLS, and hence are mainly dependent on plasma glutamine availability in situations (A) and (B).
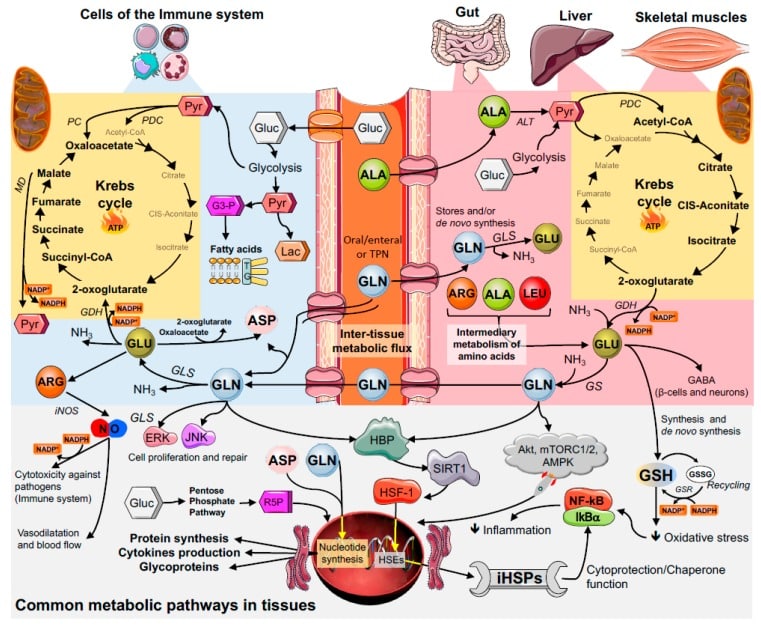
Figure 3: Glutamine inter-tissue metabolic flux starting in skeletal muscle, liver, and gut continues in the immune cells. Abbreviations: Glutamine, GLN; glutamate, GLU; aspartate, ASP; arginine, ARG; leucine, LEU; alanine, ALA; glucose, Gluc; pyruvate, Pyr; pyruvate dehydrogenase; PDC; pyruvate carboxylase, PC; malate dehydrogenase, MD; glyceraldehyde-3-Phosphate, G3-P; lactate, Lac; triacylglycerol, TG; ribose 5-phosphate, R5P; alanine aminotransferase, ALT; glutamate dehydrogenase, GDH; glutamine synthetase, GS; glutaminase, GLS; inducible nitric oxide synthase, iNOS; intracellular heat shock protein, iHSP; heat Shock Factor 1, HSF-1; heat shock elements, HSEs; sirtuin 1, SIRT1; hexosamine biosynthetic pathway, HBP; ammonia, NH3; glutathione, GSH; oxidized GSH, GSSG; glutathione S-reductase, GSR; protein kinase B, Akt; AMP-activated protein kinase, AMPK; mTOR complex 1 and 2, mTORC1/2, extracellular signal-regulated kinases, ERK; c-Jun N-terminal kinases, JNK; gamma-Aminobutyric acid, GABA.
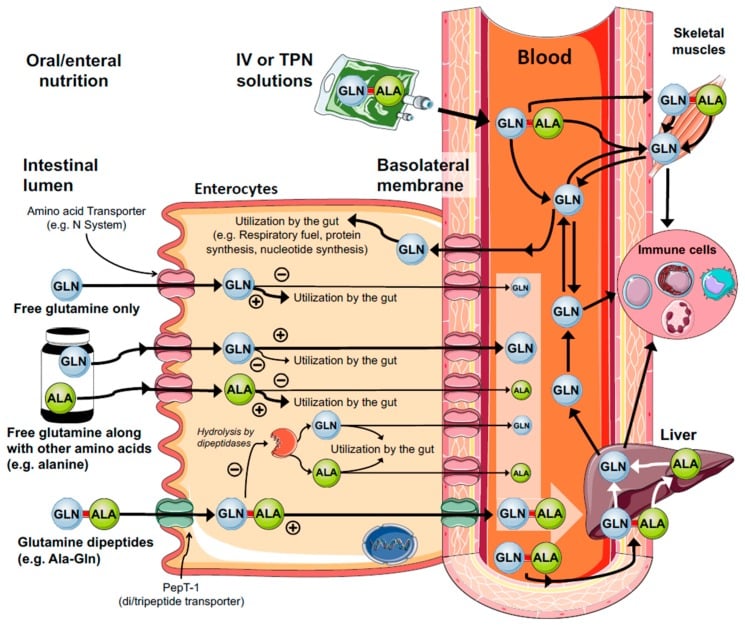
Figure 4: Mechanisms of enteral and parenteral glutamine (GLN) supply. Glutamine is an important substrate for rapidly dividing cells, such as enterocytes. This is a major site of glutamine consumption obtained from both exogenous/diet (luminal membrane) and/or endogenous glutamine synthesis (basolateral membrane). Free glutamine supplementation is mainly metabolized in the gut and poorly contribute to glutaminemia and tissue stores. On the other hand, glutamine dipeptides (e.g., Ala-Gln, Gly-Gln, Arg-Gln) escape from the gut metabolization and quickly supply glutamine to the plasma and target tissues. This effect is mainly attributed to the oligopeptide transporter 1 (Pept-1) located in the luminal membrane of the enterocytes.
similar articles
- Glutamine Metabolism and Its Role in Immunity, a Comprehensive Review.
- Glutamine-enriched enteral nutrition in very low birth weight infants. Design of a double-blind randomised controlled trial [ISRCTN73254583].
- The clinical role of glutamine supplementation in patients with multiple trauma: a narrative review.
- Specific amino acids in the critically ill patient–exogenous glutamine/arginine: a common denominator?
- [Glutamine–its metabolic role and possibilities for clinical use].
cited by
- Resistance to immune checkpoint inhibitors in KRAS-mutant non-small cell lung cancer.
- Effects of oral glutamine supplementation on jejunal morphology, development, and amino acid profiles in male low birth weight suckling piglets.
- Seafood Discards: A Potent Source of Enzymes and Biomacromolecules With Nutritional and Nutraceutical Significance.
- Effects of Glucose Metabolism, Lipid Metabolism, and Glutamine Metabolism on Tumor Microenvironment and Clinical Implications.
- Inflammatory Bowel Disease and COVID-19: How Microbiomics and Metabolomics Depict Two Sides of the Same Coin.
references
- Grohmann U., Mondanelli G., Belladonna M.L., Orabona C., Pallotta M.T., Iacono A., Puccetti P., Volpi C. Amino-acid sensing and degrading pathways in immune regulation. Cytokine Growth Factor Rev. 2017;35:37–45. doi: 10.1016/j.cytogfr.2017.05.004. – DOI – PubMed
- Curi R., Lagranha C.J., Doi S.Q., Sellitti D.F., Procopio J., Pithon-Curi T.C., Corless M., Newsholme P. Molecular mechanisms of glutamine action. J. Cell. Physiol. 2005;204:392–401. doi: 10.1002/jcp.20339. – DOI – PubMed
- Curi R., Newsholme P., Marzuca-Nassr G.N., Takahashi H.K., Hirabara S.M., Cruzat V., Krause M., de Bittencourt P.I.H., Jr. Regulatory principles in metabolism-then and now. Biochem. J. 2016;473:1845–1857. doi: 10.1042/BCJ20160103. – DOI – PubMed
- Cruzat V.F., Pantaleao L.C., Donato J., Jr., de Bittencourt P.I.H., Jr., Tirapegui J. Oral supplementations with free and dipeptide forms of l-glutamine in endotoxemic mice: Effects on muscle glutamine-glutathione axis and heat shock proteins. J. Nutr. Biochem. 2014;25:345–352. doi: 10.1016/j.jnutbio.2013.11.009. – DOI – PubMed
- Newsholme P. Why is l-glutamine metabolism important to cells of the immune system in health, postinjury, surgery or infection? J. Nutr. 2001;131:2514S–2523S. doi: 10.1093/jn/131.9.2515S. – DOI – PubMed
additional articles

Which is the Best Liver Supplement for Fatty Liver?
Are you experiencing fatigue, abdominal discomfort, or unexplained weight gain? These could be signs of a fatty liver, a condition affecting millions worldwide. With lifestyles often characterized by poor dietary choices and sedentary habits, the prevalence of fatty liver disease

How Can I Detox my Liver in 7 Days?
Have you ever felt like your body needs a reset? Perhaps you are experiencing low energy levels, bloating, or just a general sense of malaise. If so, your liver might be trying to tell you something. Could a 7-day liver

What It Takes to Choose the Best Liver Cleanse Supplements?
In a world inundated with toxins and stressors, our liver, the unsung hero of detoxification, often bears the brunt. It’s no wonder many are turning to liver cleanse supplements to support this vital organ. But how do you navigate the
Supplements to Alleviate Fatty Liver Woes- Do You Concur?
Fatty liver, a prevalent health concern, often goes unnoticed until symptoms manifest. Addressing these symptoms and considering the incorporation of supplements can be pivotal in promoting liver health. But how effective are supplements for fatty liver, and what key nuances
Unlocking Liver Wellness: Choosing the Best Liver Detox Supplement
Detoxifying your liver is a crucial step towards maintaining overall health, especially in a world where environmental toxins and poor dietary choices are prevalent. But how do you embark on this journey, and is opting for a liver detox supplement

Nurturing Liver Health: Exploring Fatty Liver Medicine
Fatty liver, a prevalent health concern in the contemporary world, necessitates a strategic approach for long-term well-being. Let’s delve into the intricacies of fatty liver, understand the considerations for effective management,. It is crucial to explore the best possible scenarios